Boost productivity
Connect. Automate. Solve.
Centralize your work, processes, and data with the united solution of Life Sciences productivity apps for sales, field, medical, marketing, events, and service.
Enrich user experiences with complete, cohesive, connected working to drive exceptional customer experiences and operational agility using Omnipresence’s diverse user personas, rich business applications, and Microsoft’s productivity solutions, like Copilot, Outlook, Teams, and Office.
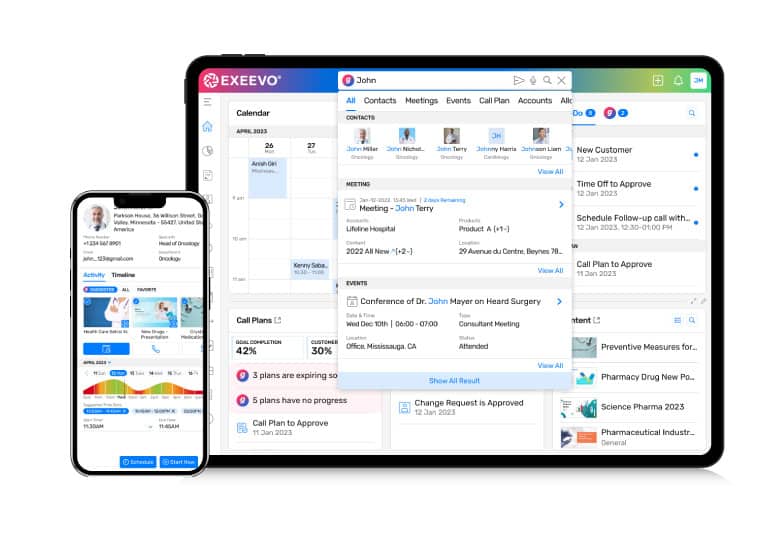
Omnipresence Copilot Artificial Intelligence
Omnipresence Copilot has reliable, scalable, and trusted AI natively integrated into our productivity apps solution-wide. Leverage AI alongside your customer data to craft personalized, predictive, and generative AI experiences that align with your business needs. Omnipresence Copilot brings conversational AI and an advanced reasoning engine to all users, enhancing operational effectiveness.
Conversational AI Assistance
This advanced feature is an intuitive assistant that engages users in natural, real-time conversations. By understanding and processing user queries, it can swiftly fetch information, offer recommendations, and automate routine tasks.
This AI assistant allows users to focus on strategic tasks and minimize administrative burdens. Because it can learn from interactions, it continuously improves, becoming an increasingly valuable asset in smoothing daily tasks to drive efficiency organization-wide.
Advanced Reasoning Engine
Enabled with deep learning and pattern recognition to analyze vast amounts of data, the advanced reasoning engine can understand the data context, including trends, outliers, and potential correlations, to provide actionable insights and recommendations and optimize workflows and processes.
Users can anticipate market changes, identify new opportunities, and respond to challenges with greater agility. This proactive approach enhances productivity and empowers users to make informed decisions that drive effective outcomes.
Empower sales teams and accelerate revenue
Make selling easier with AI automation and real-time insights built into the sales workflow
![[EXEEVO] Event Management for Life Sciences - FInal CTA](https://exeevo.com/wp-content/uploads/2024/03/Final-CTA.png)
- Predictive analytics employs AI to forecast sales trends, allowing proactive strategic adjustments.
- Automated lead scoring prioritizes potential leads using automated scoring to maximize sales efforts.
- Personalized recommendations deliver customized engagement strategies to improve interaction effectiveness.
- Real-time insights offer instant access to essential data for making informed, rapid decisions.
- Workflow and process automation streamlines daily tasks, from data entry to reporting, enhancing operational efficiency.
- Automated sales cycles speed up the entire sales process, from initial contact to closing, with systematized actions and reminders.
- Call summaries automatically generate concise summaries of sales calls, capturing key points and action items.
- Personalized close plans craft tailored strategies for closing deals, enhancing the likelihood of success.
- Prospect and account research provides thorough, AI-driven insights into prospects and accounts, equipping sales teams with necessary background information.
- Deal insights analyze deal dynamics to offer strategic advice and improve negotiation tactics.
- Conversation insights gather and analyze conversation data to refine communication strategies and improve outcomes.
Powerful Edge Analytics
Benefit from embedded analytics across all mobile device types that enable life sciences organizations to move beyond simple metrics and charts to hyper-efficient customer insights.
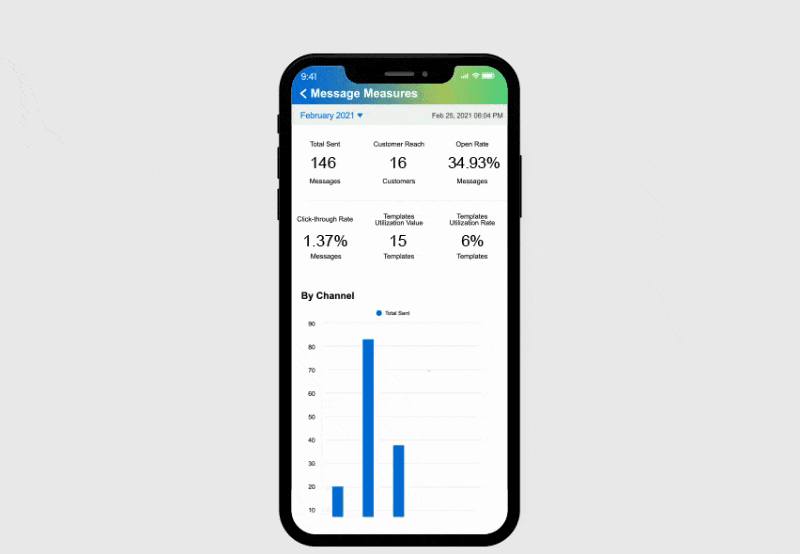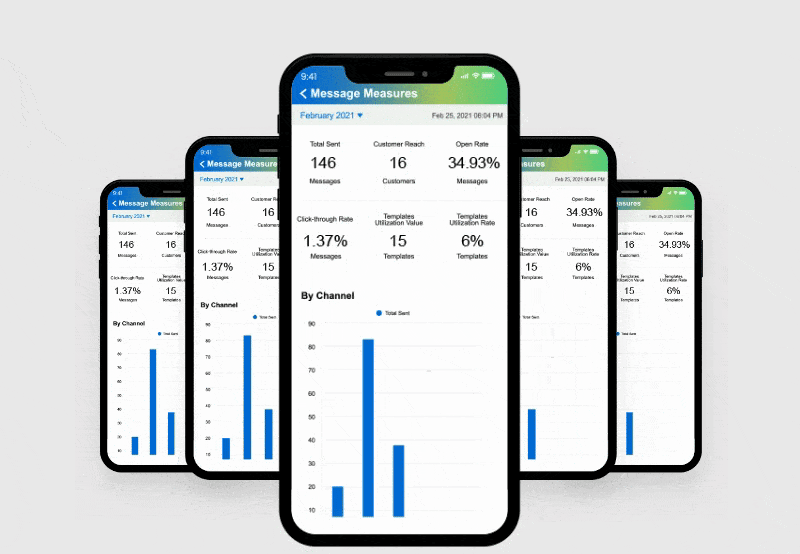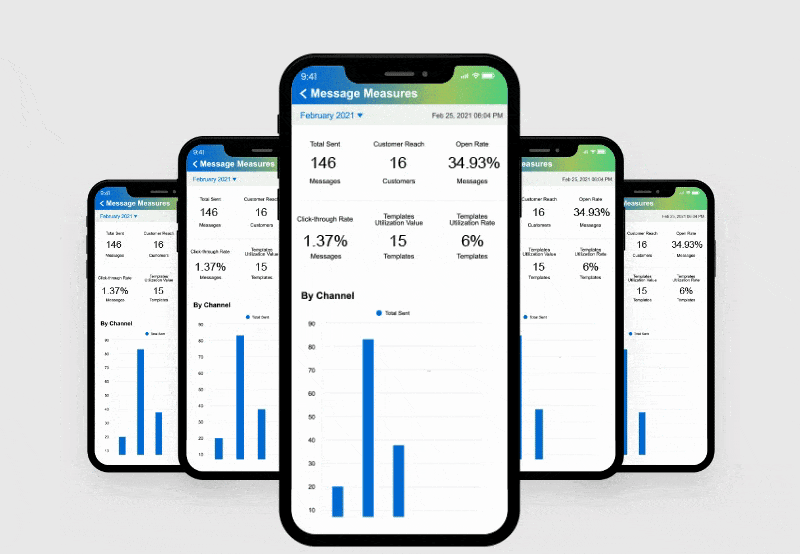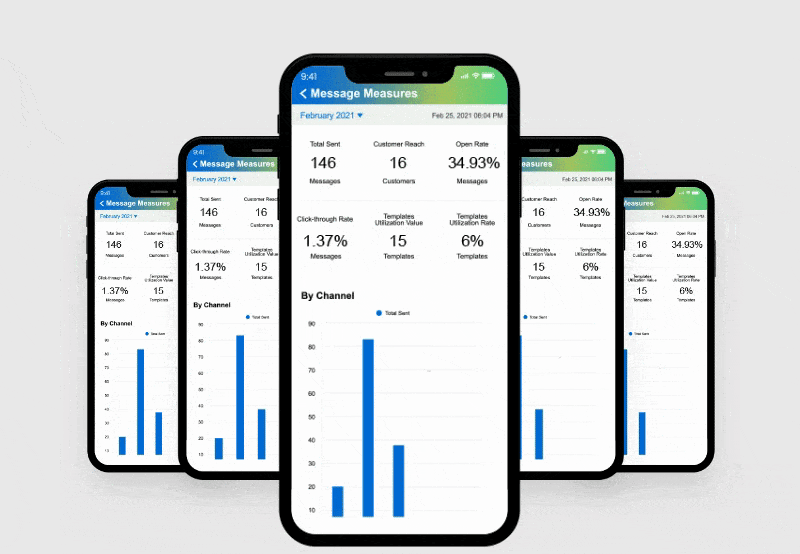
Omnipresence Copilot GO APP
The GO app is a comprehensive mobile version of Omnipresence Copilot CRM. It enhances sales efficiency and effectiveness, allowing sales reps to manage their sales activities, access vital dashboards, and retrieve files effortlessly from anywhere.
GO has features like conversational AI and voice-to-text capabilities, ensuring you can work smarter and stay connected to critical data and insights regardless of location.
The GO app seamlessly integrates the full power of Omnipresence Copilot into your mobile device, facilitating a smooth device-agnostic transition from desktop to mobile use.
GO offers advanced tools such as:
- a sales tracker for relationship tracking and analytics
- a streamlined user interface, and comprehensive sales dashboards.
- sales visit verification
- geolocation for directions
- robust lead tracking and nurturing systems.
- a built-in sales dialer and self-service portal that enhances lead generation and management.
The GO app provides personalized navigation and intelligent AI-powered recommendations to help you make informed decisions. Its intelligent analytics feature smart, contextual dashboards that visualize business metrics, spot opportunities, predict outcomes, and offer prioritized, AI-driven recommendations.
The GO app is your single solution for managing all CRM activities, offering you a powerful tool to help you work smarter, not harder.